When Patients Shape Access: Enabling Patient Access Advocacy
Pharma's role in strengthening Patient Access Advocacy

In recent years we have seen two simultaneous “push and pull” trends around patient engagement in medicines research and development.
The first is that patients have become significantly more empowered and have “pushed” for more involvement. With greater access to information has come higher health literacy, stronger collective organization, and more confidence to challenge decision-makers and demand inclusion in decisions that affect them.
At the same time patient engagement has also been “pulled” by the system, with stakeholders realizing the benefits it brings. Patient data, insights, and engagement can help regulators to better assess benefit and risk, HTA bodies and payers to assess value beyond clinical endpoints, and policymakers and industry to develop more impactful solutions and build public trust.
Our recent Ready 4 the Future? report highlighted how patient involvement has become more widespread, structured and influential, with growing recognition that patient experience data (PED) and lived experience must play a central role in shaping decisions.
Nowhere is this more important than in access to medicines.
Why access – and why now?
The access landscape is being reshaped by powerful and, at times, conflicting policy forces. In the United States, mechanisms such as Most Favored Nation (MFN) fundamentally alter how prices, incentives, and launch decisions interact across markets, amplifying the global consequences from policy changes in individual countries, leading to global impact. In Europe, the revised General Pharmaceutical Legislation (GPL) seeks to accelerate and harmonize access, explicitly raising expectations that medicines reach patients faster and more equitably across Member States.
Together, these dynamics create real tension between global value preservation and local access objectives. Tensions that are already visible in delayed launches, staggered availability, or the absence of new therapies in certain markets. Patient organizations have cautioned that shaping reforms to access processes and systems must be assessed based on their real world consequences. As the European Patients’ Forum has stated in the context of the EU pharmaceutical legislation, “it is essential to get this right now, ensuring that every measure is assessed through the lens of its direct impacts on patients and public health.” 1
In this environment, Patient Access Advocacy is emerging as a critical capability for patients and patient advocacy groups (PAGs) as patients are often the only stakeholders able to articulate the real‑world consequences of access decisions in a way that resonates with both policymakers, payers, and the public.
What is Patient Access Advocacy?
Patient Access Advocacy can be broadly defined as the organized, independent effort by patients and PAGs to influence healthcare systems, policies, and decision‑making processes to ensure that access decisions reflect real-world needs, lived experiences and meaningful outcomes. Importantly, Patient Access Advocacy is not about product promotion or price lobbying. It is specifically focused on shaping access-related decisions that ultimately affect patients.
Across access policy, HTA and reimbursement, Patient Access Advocacy operates along two complementary mechanisms:
- Generating patient‑relevant evidence that shapes how value is assessed.
- The direct participation of patient representatives in formal decision‑making processes.
Both mechanisms are essential: patient relevant evidence defines what matters, while direct participation ensures this evidence is taken into account in access decision making.
From participation to impact
For Patient Access Advocacy to be effective, it needs to influence how access is governed (access policy), how value is assessed (HTA and reimbursement), and what evidence is considered relevant for decision‑making in the first place.
Evidence generation: Patient Access Advocacy influences access outcomes by shaping what evidence is generated and whether it is considered relevant for access processes and decision-making. Beyond traditional clinical endpoints, patients and PAGs help identify outcomes that reflect meaningful real‑world benefit – such as symptom burden, functional ability, quality of life, and treatment experience – particularly where conventional endpoints fail to capture the lived impact of disease. This directly affects access decision‑making. A review of oncology HTAs across agencies (NICE, HAS, IQWiG, NHS Scotland) found patient-reported outcome (PRO) data used in about two‑thirds of assessments, with payers noting that good post‑progression PROs can have an “immediate impact” on decision making, particularly around perceived benefit and risk.2 In parallel, PAGs increasingly generate independent, patient‑led evidence – such as burden‑of‑illness studies, patient‑preference research, and qualitative experience data – to contextualize clinical results and inform access discussions, particularly in rare diseases and other settings with limited trial evidence.
Access policy: Patient advocacy is increasingly shaping policy and system frameworks – meaning patient input is contributing to the fundamentals around how access occurs, not just individual decisions. In Singapore, patient engagement is now a formal part of HTA, influencing which technologies are assessed, how value is defined, and funding decisions. Structured mechanisms, including a national Consumer Panel, enable patients to inform priorities, contextualize clinical data, and highlight unmet needs. This has increased the use of patient input in HTAs, improved decision relevance and transparency, and driven a shift toward recognizing patient insights alongside clinical and economic evidence.3 In England, patient involvement is now fully institutionalized into every stage of the HTA process – from topic selection and scoping to evidence submission, committee deliberation and consultation. Lived experience is often considered alongside clinical trial data cost-effectiveness models.7
HTA and reimbursement: Despite progress, the greatest opportunity for impact remains within HTA – not only because this is the stage that is most crucial to patient access, but because it is where the impact of patient input has yet to be fully realized. A 2025 comparative analysis of 41 HTA bodies found that “many HTA systems include patient participation,” but levels remain modest and heterogeneous.4 While there is growing recognition of patient‑centered evidence (PROs, patient preference studies, qualitative experience data), there is large variation in how systematically it is requested, appraised and weighted versus traditional clinical and economic data. And while most European systems now involve patients directly in the assessment in some way (e.g. written submissions, interviews, committee participation), the form and influence of that involvement vary widely by country.
Nevertheless, there are examples where patient input has directly influenced HTA outcomes. In Germany, reassessment of daratumumab for multiple myeloma incorporated patient-relevant outcomes including symptoms and quality‑of‑life, leading to a higher added-benefit rating despite a lack of statistical difference on overall survival.5 In England, NICE granted in a re-evaluation routine access to elosulfase alfa– a treatment for an inherited rare and progressive metabolic disorder– with patients’ lived experience playing a strong role.6
New frameworks such as the Joint Clinical Assessment (JCA) in Europe introduce mandatory patient involvement at the clinical assessment stage, forming a foundational input for subsequent national HTA and reimbursement decisions. Questions remain, however, about whether patient engagement in JCA will be sufficiently meaningful in practice.7
The challenge for pharma
Despite growing recognition of Patient Access Advocacy, many pharma organizations still struggle to translate intent into impact. Questions remain around how to engage PAGs consistently, how to support them without compromising independence, and how to prioritize efforts across markets with very different access systems.
At the same time, PAGs are highly heterogeneous. Some are established and well-resourced and already active in complex access discussions; others are earlier in their journey and require foundational capability building before they can meaningfully engage.
We propose a PAG Access Readiness Archetyping Model to help pharma teams better understand and support PAGs.
This model plots PAG Access Advocacy maturity against country-level system maturity.
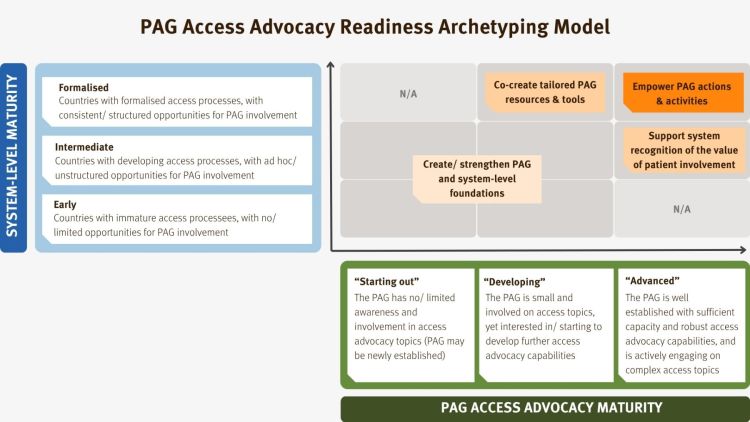
In some markets, opportunities for patient involvement in access processes remain limited. In these contexts, efforts may need to focus first on raising awareness among healthcare system stakeholders and advocating for greater inclusion of the patient voice.
Enabling impact: tailoring support with the Patient Access Advocacy Framework
Based on PAG readiness, pharma can take targeted actions to strengthen Patient Access Advocacy. Pharma can add significant value by funding and co‑creating infrastructure and skills across the following six domains:
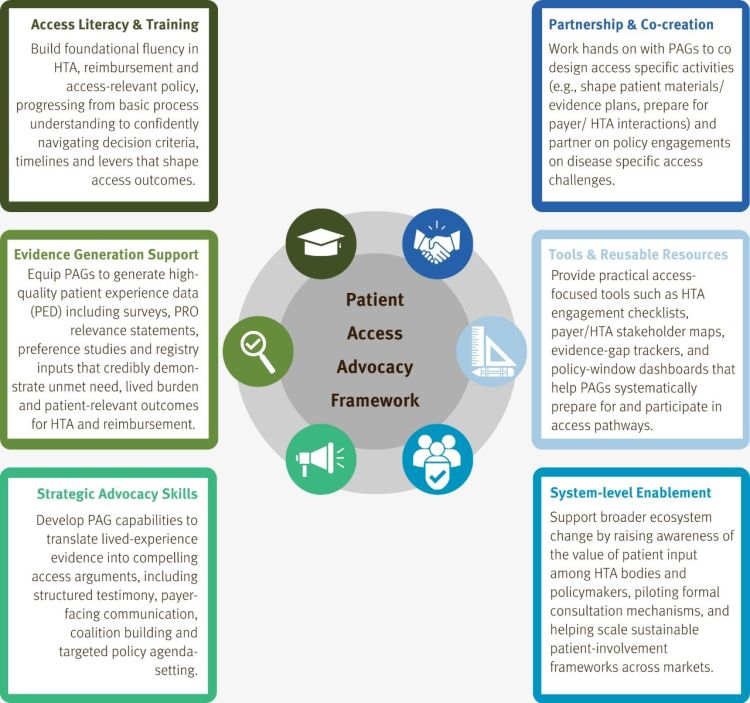
Access literacy and training: Courses from organizations like EUPATI and the European Capacity Building for Patients (EUCAPA) project as part of the EU4HEALTH initiative show that structured HTA education for patient leaders is feasible and impactful: after the first EUPATI expert course, the proportion of course participants advising HTA/reimbursement agencies doubled (4% to 8%).8 EUCAPA – organized by a three‑partner consortium led by EURORDIS and including the European Patients’ Forum and UMIT Tirol – provided multi‑level training for patient experts to contribute to JCA, focusing on HTA methods, processes, and stakeholder roles.9 Pharma can work with both initiatives to map competencies: co‑fund scholarships and course fees so PAGs can benefit both from EUPATI’s medicines‑R&D training and EUCAPA’s HTA‑specific modules.
Evidence generation support: Pharma can play a crucial role in supporting PAGs to contribute to evidence generation, including co-creation of research questions and endpoints, validating what outcomes actually matter to patients (e.g., quality of life vs. biomarkers) and identifying real-world barriers to treatment (e.g., travel, time, mode of administration). Increasingly, PAGs design surveys and qualitative studies to inform HTA submissions and access strategy, including structured patient‑experience studies and burden‑of‑illness work.10 Pharma can support PAGs in co‑creating clinical trials by engaging them early and formally in protocol design, including the identification of research questions, eligibility criteria and endpoints that reflect what truly matters to patients. This works best when companies build PAG capabilities (basic trial and endpoint literacy), compensate and structure their involvement through advisory boards or working groups, and then visibly carry their input through into PRO selection, evidence plans and regulatory/HTA documentation.
Strategic advocacy skills: HTA‑facing guidance stresses that patient input is most useful when it clearly links lived experience to unmet need, value drivers and trade‑offs, not just narrative testimony.11 Pharma can support by organizing workshops and coaching on how HTA thinks, including PICO framing, comparators, endpoints, utilities, and budget impact, and through co-development of key messages and value stories.
Partnership and co-creation: EUPATI guidance explicitly recommends close cooperation between patient organizations, regulators, HTA bodies, academia and industry, and notes that experience to date shows such partnerships increase transparency, trust and the quality of evidence.12 Pharma can support this collaboration by engaging PAGs early in submission scoping and evidence planning, working jointly to define which outcomes, impacts, and trade‑offs matter most to patients, and to ensure that value is articulated in a way that is relevant for decision-making by access bodies.
Tools and reusable resources: Many high quality resources exist to support PAGs in access initiatives, most notably EUPATI’s toolbox providing guidance for patient involvement in HTA, and the EUCAPA project. In addition specific resources are available on selected topics, e.g. IAPO’s UHC Compass, which provides practical support for PAGs on how to advance universal health coverage (UHC) in their communities. However, opportunities still exist for pharma to co-create very tailored open‑access toolkits with PAGs focused on specific conditions or access situations, including HTA engagement checklists (what to do before/during/after an appraisal) and stakeholder mapping for each country.
System-level enablement: It has been suggested that demonstrating the value of patient contributions (e.g. through retrospective impact analyses) is key to embedding involvement structurally in HTA.13 Pharma can support this by conducting research (potentially alongside HTA bodies and academia) to document how patient input has changed HTA questions, deliberations or recommendations.
Now is the time for Patient Access Advocacy
Patient engagement has come a long way, from inclusion to creating real impact. The next step is to fully embrace Patient Access Advocacy: patient‑led, evidence‑informed, and system‑focused involvement in how access decisions are designed and made. This means supporting patients and PAGs to actively shape access discussions, grounded in robust patient‑relevant evidence and a clear understanding of how healthcare systems function.
This moment is critical. With increasing pricing pressure, tighter budgets, and accelerating policy change across markets, access is no longer determined by clinical and economic value alone. It also depends on whether health systems recognize and can act on what matters to patients: their needs, priorities, and lived experience. Delayed access has far-reaching consequences that extend beyond individual patients, affecting societal participation, workforce sustainability, and economic resilience.
For pharma, this reality requires a shift away from one‑off engagement towards a more structured, capability‑led approach to Patient Access Advocacy. It means understanding where PAGs stand today, where and how patient input is embedded in access processes, and what targeted support is needed to enable meaningful, sustained contributions over time.
Companies that invest early in Patient Access Advocacy – by strengthening patient capability, supporting the generation of patient‑relevant evidence, and embedding patient perspectives across access policy, HTA, and reimbursement – will be better positioned to navigate policy uncertainty and enable sustainable access. Ultimately, the companies that succeed will be those that recognize Patient Access Advocacy not as an adjunct to access strategy, but as a strategic force shaping how value is defined, evaluated, and delivered.
Download the entire article here (pdf)
References
1 European Patients’ Forum (EPF). EPF Statement on the Pharmaceutical Legislation, 2025.
https://www.eu-patient.eu/news/latest-epf-news/2025/epf-statement-on-pharmaceutical-legislation/
2 Brogan, A. P., DeMuro, C., Barrett, A. M., D'Alessio, D., Bal, V., & Hogue, S. L. (2017). Payer perspectives on patient-reported outcomes in health care decision making: Oncology examples. Journal of managed care & specialty pharmacy, 23(2), 125-134.
3 Pearce F, Tan PT, Liow J. Singapore: From stakeholder mapping to continuously improving patient participation in HTA. In: Facey KM, Holtorf AP, Single AN, eds. Patient Involvement in Health Technology Assessment. Health Informatics. Cham, Switzerland: Springer; 2026. doi:10.1007/978-3-032-11284-2_23
4 Puebla M, Korrodi-Gregório L, Trentin L, De Maria A, Blanqué N. A comparative analysis of patient participation in health technology assessment systems worldwide: trends and practices. Front Public Health. 2025;13:1693886. Published 2025 Oct 31. doi:10.3389/fpubh.2025.1693886
5 Institute for Quality and Efficiency in Health Care (IQWiG). Contributing the patients’ perspective. Press release. Published 2022 Jan 3. Accessed April 9, 2026. https://www.iqwig.de/en/presse/press-releases/press-releases-detailpage_57992.html
6 https://mpssociety.org.uk/latest/nice-approves-life-changing-medication-for-treating-mps-iva-morquio
7 Arcà E, Barlassina A, Eze A, Strammiello V. Enhancing Patient Engagement in HTA: Using Consensus Research to Overcome PICO Scoping Challenges Under the EU HTAR. J Mark Access Health Policy. 2025;13(2):27. Published 2025 Jun 2. doi:10.3390/jmahp13020027
8 Hewitt C. Empowering patient involvement in HTA through effective education: the impact of EUPATI. Remap Consulting. Published April 6, 2018. Accessed April 8, 2026. https://remapconsulting.com/hta/empowering-patient-involvement-in-hta-through-effective-education-the-impact-of-eupati/
9 EURORDIS-Rare Diseases Europe. EURORDIS launches EUCAPA. Published March 7, 2023. Accessed April 14, 2026. https://www.eurordis.org/eurordis-launches-eucapa/
10 British Healthcare Business Intelligence Association (BHBIA). Patient focused research to inform regulatory and HTA assessment. Published June 10, 2021. Accessed April 8, 2026. https://www.bhbia.org.uk/events-courses-webinars/patient-focused-research-to-inform-regulatory-and-hta-assessment
11 de Wit M, Guillemin F, Grimm S, et al. Patient engagement in health technology assessment (HTA) and the regulatory process: what about rheumatology? RMD Open. 2020;6(3):e001286. doi:10.1136/rmdopen-2020-001286
12 Hunter A, Facey K, Thomas V, et al. EUPATI guidance for patient involvement in medicines research and development: health technology assessment. Front Med (Lausanne). 2018;5:231. doi:10.3389/fmed.2018.00231
13 Arcà E, Barlassina A, Eze A, Strammiello V. Enhancing Patient Engagement in HTA: Using Consensus Research to Overcome PICO Scoping Challenges Under the EU HTAR. J Mark Access Health Policy. 2025;13(2):27. Published 2025 Jun 2. doi:10.3390/jmahp13020027