All PROs, no cons? Why patient-reported outcomes can ensure the patient voice is fully heard in access decisions
Despite growing acceptance around the importance of patient input and evidence in healthcare decision-making overall, there still seems to be great inconsistency when it comes to the acceptance of patient evidence in access decisions. Executive Insight and merakoi have conducted surveys canvassing payer, patient and pharma opinions on patient evidence in health technology assessment (HTA)/access decision-making.

“Patients sit in the meetings, but the transcripts show that they seldom or never speak.”
- Payer, France
“There is a large ignorance by the regulators and payers of the impact of a disease on a patient.”
- Patient, Italy
These two quotes – from recent surveys conducted by Executive Insight and merakoi canvassing payer, patient and pharma opinions on patient evidence in health technology assessment (HTA)/access decision-making – shine a light on some of the challenges around amplifying the patient voice in this area. Despite growing acceptance around the importance of patient input and evidence in healthcare decision-making overall, there still seems to be great inconsistency when it comes to the acceptance of patient evidence in access decisions.
In the Executive Insight survey, only 38% of payer respondents stated that patient evidence is either often (13%) or always (25%) considered in HTA/ access decisions in their country. Meanwhile, 85% of patient experts in the merakoi / Executive Insight survey believed that patient evidence is not adequately considered in decision-making.
Why is this the case? After all, use of patient evidence and engagement has proven to be effective in other areas of drug development. In 2018, the Economist Intelligence Unit compared 4,000 clinical trials with significant patient-centered elements with 20,000 traditional trials – 87% of the patient-centric trials had positive results compared to 68% of traditional trials. The same study revealed that patient-centric trials took on average three months less to recruit participants compared to traditional trials.
But when it comes to access decisions, patient evidence is neither considered, nor given the same weight as other evidence. Of the payers in the survey who stated that HTA/access decision-making in their country considers patient evidence, the vast majority (86%) agreed that it has lower weighting compared to traditional clinical endpoint evidence.
But as EUPATI has stated, “Clinical effectiveness measures cannot tell us how a patient feels or functions, or what they want to achieve from a treatment.” It is vital that evidence that is most meaningful to patients is captured and reflected in key decisions.
Applying science to the patient voice
The key is in quantifying mutually-acceptable, objective measures of patient evidence. As a 2019 editorial in Patient stated, “Patient passion is important, but it must be combined with a more dispassionate, scientific understanding”.
Patient-reported outcomes (PROs) – health outcomes directly reported by the patient who experienced them – are a key method of quantifying and objectivizing the patient experience and, as EUPATI puts it, “provide a patient perspective on a disease/treatment that might not be captured by a clinical measurement but may be as important to the patient.”
There has certainly been some progress in this area. The importance of PRO data is becoming more widely recognized by payers worldwide as a key component of their decision-making process. Today, an increasing number of oncology medications, for example, enter the market with product labeling claims that contain PRO data, meaning payers already need to familiarize themselves with the opportunities associated with PRO evidence when making coverage decisions.
Some HTA agencies – such as NICE in the UK, IQWiG/ G-BA in Germany, CADTH in Canada and HAS in France – are expanding their focus on patient-centred outcomes and patient experience data. In the UK for example, proposed process changes by NICE include elements to more systematically incorporate the patient voice and an acknowledgement that PROs “can capture important aspects of conditions and interventions” and should be “appropriately validated.”
Reaching “mainstream respect and acceptance”
However, “PROs still have not achieved mainstream respect and acceptance,” as one surveyed patient remarked. A key reason is the persistent view among many policy makers and payers that PROs are subjective and qualitative, while the HTA process is the opposite.
This is supported by the Executive Insight survey, which revealed that payers are concerned about the standards and methodologies to generate patient evidence, the quality (i.e. high heterogeneity) and representativeness of such data, and the clinical and economic significance of the observed effect from PROs.
It is true that historically PROs have sometimes been lacking in quality or robustness, especially in terms of critical methodological aspects of collection and analysis. Too often PROs are not disease-specific, have not been developed with patients, or are not really patient relevant –PROs have typically been developed by key opinion leaders who “knew their patients”, rather than from understanding the direct experiences and perspectives of patients themselves. The use of traditional PROs assessing quality-of-life in Duchenne Muscular Dystrophy (DMD) and Multiple Sclerosis (MS) are examples where PROs have not adequately captured what is meaningful for patients, including e.g. DMD: access to care, emotional effects of disease, social skills, and caregiver impact; MS: emotional and daily life impacts, cognitive function, pain, recall period and scoring scale.
Meanwhile, the patient experts in the merakoi / Executive Insight survey cited other barriers to the use of patient evidence, including difficulty integrating patient evidence with other types of evidence and a lack of payer understanding of, and familiarity with, patient evidence.
The good news is that all three key stakeholder groups – patients, pharma and payers – agreed that patient evidence should be more systematically included in HTA/access decision-making, but how can this be achieved in a way that overcomes these diverging concerns?
Educate, create, facilitate
Early co-creation is key, making the integration of patient evidence becoming more systematic.
We have identified three strategic opportunities – which pharma can drive in partnership with patient communities – to manage this evolution. There is some ongoing work in all three areas, which pharma companies can help to focus and build upon. It is essential that pharma companies first have the right capabilities in place internally as a foundation.
1. Educate: Raising awareness and building trust in patient evidence at the HTA/access level and facilitating this through policy efforts on the value of patient evidence.
- Several organizations, including the European Patients Forum, Eurordis, EFPIA and PhRMA have been active with initiatives in this area, providing an excellent foundation to build upon and amplify.
2. Create: Early co-creation of disease-specific patient reported outcome measures (and subsequently enhancing pivotal trial design to incorporate these).
- One example is the SMA Independence Scale (SMAIS), which measures the amount of assistance required by people living with spinal muscular atrophy (SMA) to perform typical daily activities, such as getting dressed or self-feeding. This PRO was developed with the SMA community and provides an example of what can be achieved with early co-creation.
3. Facilitate: Exploring new ways to involve, train and build the capabilities of patients and patient organizations to contribute in a meaningful way.
- A range of high-caliber multi-stakeholder training materials exist, including those developed by EUPATI and PFMD, which can be utilized. There is a need for new tailored training and materials to be developed at the disease-specific level, which help patients articulate their situation in a way that is meaningful for payers.
There also needs to be better measurement systems in place to track the level of patient and societal benefit a new treatment delivers and how PROs and patient evidence improve the quality of access/HTA assessments and decisions. This would help to incentivize all stakeholders towards realizing the strategic opportunities.
Moving towards earlier, more systematic patient involvement and evidence
To ensure this progress continues across more HTAs/access decision-makers, all key stakeholders – pharma, payers and patients alike – have a role to play. Genuine co-creation and open lines of communication are vital. If all three align at an early stage to agree what meaningful patient evidence looks like and how to incorporate it into reviews, it can work, although elements of healthcare system policy change are likely required to systematically integrate patient evidence into the HTA/access decision-making process.
The below image shows the potential roles of each key stakeholder.
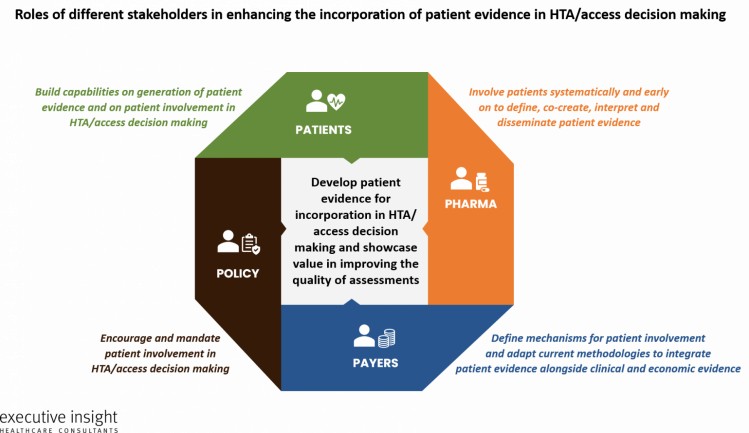
All of the patient experts in the merakoi / Executive Insight survey agreed that pharma needs to play a key role in facilitating the incorporation of patient evidence in decision-making. This could include ensuring PROs are developed early on with patients to be integrated into clinical trials, gaining patient input early in trial design, validating patient evidence methodologies and publishing patient evidence together with clinical data.
Realistically, as pharma are the ‘owners’ of the product lifecycle, they need to be in the driving seat when it comes to proactively setting up multi-stakeholder initiatives and alignment. As one US payer put it: “They fund and conduct the clinical trials - in the absence of pharma, it would be difficult to see a path forward where another entity will step in to generate this type of evidence”.
It’s vital that collection of patient evidence happens early. EUPATI guidance states that the pharmaceutical industry should “strive to involve patients early in medicines development, preferably before the clinical development phases.” After this point, “many key decisions about a medicine have already been taken and cannot be reversed”. For PROs, this means developing and validating disease-specific measures early on for use in clinical trials across the same disease area, as an industry standard, rather than seeing PROs as a “one-off” measure for a particular clinical trial.
The majority of payers (69%) in the survey also supported the pharmaceutical industry’s active role in facilitating the incorporation of patient evidence, although some have concerns around lack of transparency, conflict of interest and biased collection/interpretation of evidence.
The onus is on payers meanwhile to develop models and methodologies that can integrate multiple evidence inputs (clinical, economic, patient etc.) and come to balanced decisions. There has to be the will and the means to ensure patients have a seat at the table and that their input is properly integrated and considered in access decision-making.
From the patient perspective, the focus should be on maximizing their voice through training and capability building to become true patient experts, able to contribute objectively and meaningfully during HTA/access meetings and co-creation of PROs with pharmaceutical companies on what is meaningful to patients and how this can be optimally measured.
Ensuring patients are involved earlier to develop more meaningful patient evidence for consideration at HTA/access decision-making will benefit all stakeholders. For pharma, clinical programs will be focused on what really matters to patients, increasing the likelihood of developing truly impactful treatments and meeting unmet needs. For payers, they will have a more complete picture of what constitutes value to patients, assisting with difficult decisions on spending priorities. And for patients, they will be empowered and ultimately benefit from improved access to potentially transformational medicines.
For all three, the systematic integration of patient evidence into HTA/access decision-making provides an opportunity to break down silos and build long lasting relationships so at last, when patients talk, they are truly heard.
You can find the original article published in PME magazine December 2021 here.
You can find an entire whitepaper on this topic here: https://www.executiveinsight.ch/en/insights/publications/patient-evidence-hta-does-it-matter













