Navigating the maze of modern Medical Affairs stakeholder engagement
The fundamentals of implementing a new Medical Affairs engagement model – and why it may be essential

Medical Affairs. The definition of this term has changed almost beyond recognition in recent years.
A perfect storm of innovation in science, technology and data is transforming the healthcare environment and shifting how the pharma industry develops and communicates about its products. Information channels are changing. Demonstration of value and patient outcomes is everything. Demand for scientific knowledge and insights is growing. And with a wider range of stakeholders influencing purchasing and other healthcare decisions, pharmaceutical companies need to interact with complex ecosystems of stakeholders, not just individuals in silos.
It is against this backdrop that Medical Affairs has emerged as the “third strategic pillar” of the pharmaceutical industry alongside R&D and Commercial, with a vital role to play in helping companies navigate through this complex environment. Its detachment from commercial elements, focus on science and patient outcomes and ability to provide a credible interface with external stakeholders means Medical Affairs is uniquely positioned to take on this leading and strategic role.
From primarily liaising with individual healthcare professionals (HCPs) in the past, Medical Affairs teams must now become partners across the entire health ecosystem, collaborating with health care teams and health systems to achieve more meaningful (and measurable) outcomes.
So, what does this evolution look like in practice?
Mind over matter
The biggest essential shift within pharma companies is one of mindset. The role of pharma is moving away from simply bringing products to market, towards providing holistic solutions for better patient outcomes. This requires identifying the required outcome first and working backwards from there.
Roche is one company making this transition, implementing decentralized execution across its entire Pharma organization (product development, pharma technical, commercial) that follows a common principle of outcomes-based planning.
Medical Affairs is at the heart of this evolution. A modern Medical Affairs strategy needs to be firmly rooted in setting (measurable) care improvement goals and expectations.
To make patient-centric healthcare a reality, Medical Affairs must partner with a broader range of stakeholders than before – this may be with a whole care team, a multidisciplinary healthcare management team within an institution, or with the entire institution itself, involving holistic healthcare stakeholder engagement including the institution, care providers, and care networks (see Figure 1). This represents a significant shift as traditionally, Medical Affairs worked with thought leaders and other individual healthcare professionals. But the customer engagement model is changing, and HCPs are not the sole decision-makers they used to be. Now, it is about networks, not individuals.
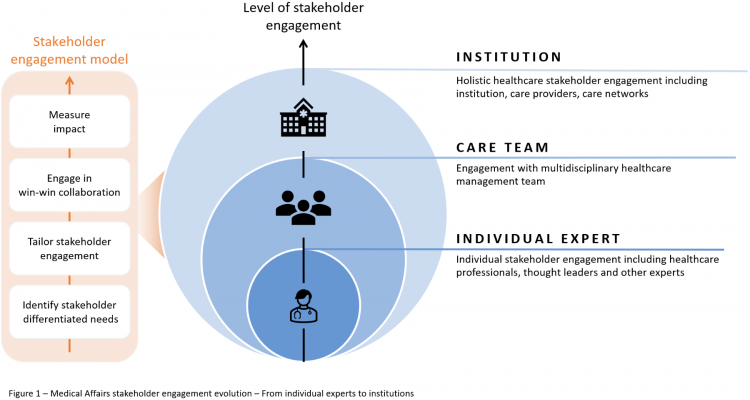
So how should stakeholders be identified in the first place? It means a shift from the traditional identification of thought leaders e.g., through publications, society memberships and so forth, to an assessment of the HCP care involvement understanding additional factors such as for example e.g., engagements in education, diagnosis rates, communication, and social media profile and so on.
Collection and analysis of quality data will require analytics expertise. Selecting and working with the right data provider to provide state-of-the art HCP identification capabilities is rapidly becoming a differentiation factor. But only by complementing data-generated stakeholder insights with inhouse knowledge such as stakeholder engagement preferences and field HCP insights can a true competitive advantage be generated.
Bigger customers, bigger goals
It is then about finding or defining win-win collaborations. Joint objective-setting is essential to identify an outcome that will fulfil an unmet need for the care team or institution – while being aligned with the pharma company strategy – and which can realistically be achieved through co-creation. As we are now at the care team / institution level, these outcomes can be quite ambitious, for example improving diagnosis rates, or patient referral rates, or establishing a new science, technology, or approach into clinical practice.
When it comes to the nature of the engagement to achieve the outcome, the Medical Affairs team should be equipped to support a wide range of activities, for example evidence collection, employee capability training, supporting guideline updates or a range of medical education activities.
For example, AstraZeneca entered into a collaboration agreement with the Massachusetts General Hospital (MGH) to pilot its AMAZE disease management platform with the goals of improving patient engagement, care-team communication and clinical outcomes while reducing healthcare costs.
Meanwhile, Bayer introduced its Alleve Home Monitoring Sponsorship Program to the Egyptian healthcare sector, working in partnership with some of the country’s top-rated eye hospitals to support Egypt’s ophthalmologists with its mobile medical software application that detects, characterizes, and/or tracks progression of visual distortion in patients with diabetic retinopathy and/or age-related macular degeneration [AMD].
The engagement approach will vary from company to company but a single point of contact, in the style of a Key Account Manager (KAM) – orchestrating activities across the broader field and internal team – will more likely generate the desired impact. Direct engagement through medical field teams and channels will build strong relationships, and independent support can equally support high-impact care improvements. A strong grant strategy, including considerations of independent medical education programs – or funding of quality improvement/performance improvement programs, often in combination with evidence generation – complements the medical engagement mix.
Made to measure
With increasingly ambitious goals, greater investment and more at stake, measurement of the impact is more important than ever. Because the impacts of these ventures are long term, spanning more than a business cycle, robust and jointly-created KPIs are vital to justify the continued efforts and maintain motivation for the external care team as well as the pharma company.
Exploring hospital specific records and outcomes data can be an excellent starting point for identifying areas to improve and track progress towards them. To ensure a tangible outcome, opportunities to generate real-world evidence data or produce publications on outcomes achieved should be considered. Such activities are very beneficial for raising the profile of the project and increasing the win-win nature of the partnership.
It can take some time to define and measure the right “outcomes” data, but it can usually be done. Medical education is an area where this has been achieved, for example via knowledge measurement. An example from the Asia-Pacific region is the industry-funded ASPIRE training program — the Asia-Pacific Initiative for Rheumatology nurse Education – designed in response to the increasing challenges experienced by rheumatologists in managing their patient caseload and recognizing the value of the nurse specialists in clinical practice. Measurement of the ASPIRE training on a participant level of knowledge, confidence, attitudes and beliefs using a before-after-control-impact survey was carried out, demonstrating a significant increase in knowledge and confidence.
The new Medical Affairs engagement model: Imperatives for success
Linked to the fundamental change of mindset within the organization is the need to bring internal teams onboard, ensuring alignment and lack of duplication. A willingness to learn and experiment within the organization is important to allow the new way of working to thrive. As with any change-management program, endorsement from senior leadership is key to success and to fostering a culture where this is feasible. At the same time, guardrails must be in place to ensure the basic governance and industry regulations are fully adhered to.
It is important that engagement is tailored to the specific needs of the partner to maximize the chance of mutual buy-in to a win-win initiative and an ongoing commitment from all parties. This requires openness and transparency by Medical Affairs teams to co-shape the partnership and co-create solutions.
There needs to be a clear delineation of roles, ensuring the right balance between joint efforts and independence. The specific roles and responsibilities of each party needs to be completely transparent.
Finally, there is a need to tailor engagement to geographical differences. The medical affairs engagement model will need to be localized, country by country, which is a new way of working for many companies. Consideration of the unique external context in each country (e.g., how care systems are structured, local regulations and policies allowing for different kinds of engagement model etc.) will be essential to success. Such an approach relies on cross-country transparency, knowledge management and consideration of country archetyping to reduce duplication, maximize success and share existing expertise and materials that are relevant to achieving the desired outcome.
Conclusion
The evolution of the Medical Affairs function is happening at a varying pace across the industry. The general trend is a move away from the traditional approach of focusing on individual thought leaders and HCPs towards more complex and networked engagement with the whole care team or institution.
Several forward-looking pharma and life sciences companies have already recognized the opportunity to forging strategic partnerships with the aim of advancing care and improving patient outcomes, supported by their products and solutions.
The real pacesetters are already fully integrated into new ways of working, implementing fit-for-purpose internal structures with engaged and fully invested leadership, and focusing on building a rich set of proprietary data and digital healthcare solutions.
But from a big picture across the whole industry, we are only at the very beginning of this journey: Companies still have the opportunity to make this new Medical Affairs engagement model a compelling point of differentiation.










