Can the patient pathway provide a route to success?
Why pharma companies need to identify barriers and co-create solutions along the patient pathway to ensure their new products reach the patients who need them

Remember the Segway?
A two-wheeled, self-balancing personal transporter launched in 2001, it was the first of its kind, very innovative and came with high expectations from its developers.
But commercially, it failed miserably. A high price tag didn’t help, but the key reason it failed was its impracticality. With most cities designed for either pedestrian or large vehicles, the Segway lacked both the infrastructure and necessary regulation to succeed. The developer simply hadn’t prepared the market for its use.
Quite literally, there was no pathway for it.
Several pharmaceutical companies can probably relate to this experience. Many have also launched first-of-their-kind, highly innovative products onto largely unsuspecting markets, with high expectations. But with healthcare systems becoming increasingly complex, with more stakeholders, multifaceted treatment protocols, and all the while budgets being stretched further and further, a more holistic approach is now needed.
It is estimated that about 40% of new medicines approved between 2004 and 2016 underperformed Wall Street’s prelaunch sales forecasts by over 20% in their first three years on market. Ultimately, this means that a lot of products are not reaching the patients who need them.
There are many examples of new therapies struggling to find their place in existing pathways. Direct-acting antivirals (DAAs) for hepatitis C infections (HCV), for example, promised to transform the treatment landscape in this area. But it has taken years of significant care structure re-organization, shifting focus from specialist centers to community-based primary care, for them to reach the numbers of patients initially expected at launch.
Barriers need to be identified at each stage of the patient pathway as early as possible so that pharmaceutical companies can work with health system partners to co-create solutions to overcome them, and thus improve care for patients in the healthcare system.
Mapping the four stages of the patient pathway
But first, it is important to define exactly what we mean by ‘patient pathway’.
Essentially, a patient pathway is the journey of patients through a health system, usually within a specific disease area, from an individual’s recognition of symptoms, leading to their first contact with a healthcare professional (often his or her GP), through to overcoming the condition or ongoing disease management.
While patient pathways will inevitably vary across disease areas, countries and jurisdictions, there are some fundamental elements that will likely always be included. Figure 1 maps out the four key stages of the patient pathway, the various stakeholders involved at each stage, and their roles.

To illustrate the point, let’s look at the example of Alzheimer’s disease (AD). With a range of innovative therapies in development, it will be critical to have an efficiently functioning patient pathway for their true impact to be realized. But currently, a variety of challenges exist at every stage:
Stage 1 – Awareness & Recognition: A lack of awareness of the early signs and symptoms of cognitive decline, combined with the complexity of stigma and the fear of AD, contribute to a delay in detection. Despite being a clear urgent unmet need, AD is not yet being prioritized enough across stakeholders to raise public awareness.
Stage 2 – Presentation & Diagnosis: Advances in diagnostic biomarker testing in the academic setting are yet to translate into clinical practice. Almost all healthcare systems face challenges in having the right infrastructure and capacity to perform lumbar punctures to obtain cerebrospinal fluid (CSF) samples for beta amyloid testing, or PET scans to measure the build-up of amyloid protein in the brain, for example. In addition, the pathway itself is often fractured through lack of standardized referral routes, and a lack of guidelines for healthcare professionals (HCPs).
Stage 3 – Initial Treatment & Coordinated Care: When an effective new treatment becomes available, there will be extra demands on healthcare systems to administer treatments, for example through infusion centers, if required. With significant fragmentation between different HCPs involved in AD care, much better coordination will also be required. Lastly, funding and affordability of such innovative treatment will build an additional barrier to broad access for patients as well as a significant new demand on healthcare systems’ budgets.
Stage 4 – Disease Management / Living with Condition: The infrastructure of many healthcare systems looks unlikely to be able to withstand the increased demand for testing required to monitor patients for safety and disease progression upon introduction of new therapies. The significant burdens on the social care system and individual patient carers will also continue to be a major factor.
Turning insights into action
Mapping out the patient pathway provides clarity on the barriers to optimal care, but it is only the first step to take if pharma companies are to effect real change in the healthcare system.
In fact, pharma can play an integral role in addressing these barriers. Example types of solution include engaging cross-stakeholder audiences, generating and disseminating data, providing tailored education, and partnering to co-create and implement targeted solutions.
In order to realize their potential role, pharma companies must adopt a structured approach to environment transformation, with buy-in from across the organization. Figure 2 provides a broad three-step approach that can be implemented to optimize the patient pathway, with modules that can be combined in a flexible way to reflect the environmental need.
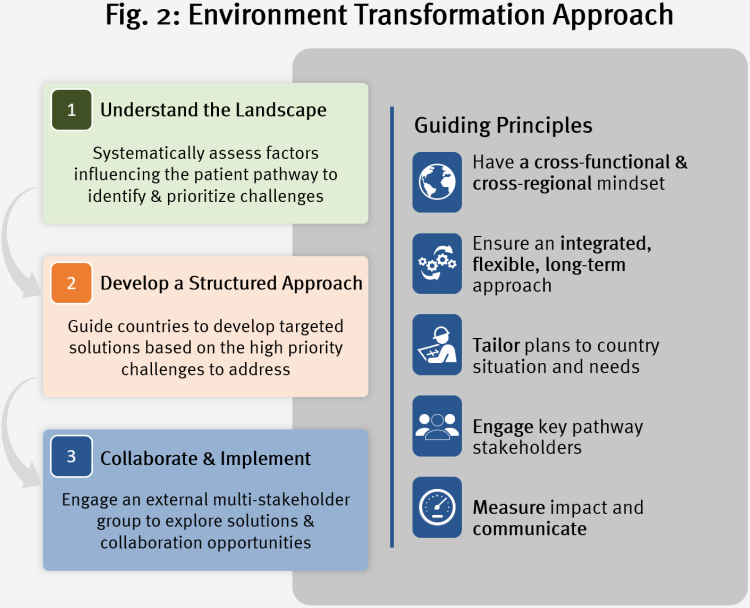
What does this look like in practice? Below are examples at each stage of the patient pathway, where pharma companies have worked with other stakeholders to identify barriers and co-create impactful solutions.
Stage 1 – Awareness & Recognition: ‘Agents of Change’ Atopic Dermatitis Challenge
Recognizing that the challenges of living with atopic dermatitis are far from skin deep, Sanofi launched a global grants initiative called ‘Agents of Change’. The program sought proposals for grassroot solutions to help solve some of the greatest challenges impacting people living with the condition. Not only did this have a direct impact on the five grant winners – which included a youth led education and awareness campaign, and anti-bullying ambassadors – but it helped to shine a spotlight on some of the real day-to-day challenges faced by those with atopic dermatitis.
Stage 2 – Presentation & Diagnosis: European Alliance for Newborn Screening in Spinal Muscular Atrophy (SMA)
The need for a swift diagnosis is perhaps nowhere more evident than in SMA, when delays can lead to an irreversible loss of motor neurons, with a loss of more than 90% of motor units within 6 months of birth. Because administration of innovative SMA products is significantly more effective before the onset of symptoms, newborn screening programs are essential for SMA, but are not always in place.
Cross-stakeholder initiatives, such as the European Alliance for Newborn Screening in Spinal Muscular Atrophy, have been fundamental in effecting change. SMA testing is now included in many countries’ newborn screening programs and the Alliance – which includes Novartis, Biogen and Roche as industry partners – has published a white paper calling for screening in all European countries by 2025.
Stage 3 – Initial Treatment & Coordinated Care: ‘Act4Biosimilars’ initiative
While biosimilars provide more affordable alternatives to biologics, access inequities are preventing many patients benefiting from them. An initiative by Sandoz called Act4Biosimilars – supported by a committee of patient advocacy leaders, healthcare professionals, biosimilar experts and industry leaders – aims to increase patient access to advanced medicines by facilitating greater approvability, accessibility, acceptability and affordability (the 4 A’s) of biosimilars. The initiative includes an Action Plan to provide the strategies, tools and activities needed across 30 target countries, supported by Country Indicator Maps to track and measure the change.
Stage 4 – Disease Management / Living with Condition: Remote care model in type 2 diabetes
A significant number of people with type 2 diabetes (T2D) do not take medication as prescribed and therefore have poor outcomes. In North-West London – where T2D costs £600 million every year and accounts for over 40% of all hospital admissions – a partnership including AstraZeneca, HUMA, Imperial College London, and Health Data Research UK piloted a new remote care model. The initiative first used risk prediction algorithms to identify the highest risk patients, who then were invited to join an intensive digital support program. The program included both in-person group consultations, to drive behavior change, medicine intensification and diet and lifestyle coaching, and a smartphone app and monitoring devices for participants to measure and report important indicators for diabetes without the need for GP consultations. The successful model is currently being rolled out across North-West London.
A pathway to the future – but which way will it take us?
Identifying and implementing solutions within patient pathways will not be straightforward, and there are common pitfalls encountered when approaching environment transformation. Perhaps the most common is a lack of cohesion throughout the organization. Due to the complex, cross-stakeholder nature of the patient pathway, environment transformation requires alignment around a common goal across functions and geographies. Without this, the company cannot hope to create meaningful change at scale. Another common pitfall is to approach transformation at the wrong time, either too late in the planning cycle to effect real change, or too early such that sufficient resources are unavailable.
Challenges in the approach will probably be most pronounced in cases where significant changes to the pathway are required – for example as more personalized and innovative medicines are integrated into healthcare systems already under pressure.
We will likely also see the tools and solutions used continue to evolve with advances in data and digital technologies such as artificial intelligence (AI) for screening. And in some cases, it may not be a case of evolving the patient pathway but changing it completely, for example with the integrated practice unit (IPUs) model.
But whether patient pathways are evolved, or fundamentally redesigned, pharmaceutical companies need to embrace this approach in any therapeutic area in which they are involved. By engaging in timely environment shaping, companies can create a win-win that ultimately results in smoother patient pathways with their product being properly utilized to improve the care of patients who need it. While this requires early engagement with stakeholders and investment in solutions beyond the pill, the potential value created for the organization and the wider population is significant.
By embracing the patient pathway, new innovative products won’t go the same way as the Segway – but will rather be a segue to better health.
You can find the original article published in Pharmaceutical Executive Magazine here: https://www.pmlive.com/pme#/reader/48972/214704
References
Newborn blood spot screening test using multiplexed real-time PCR to simultaneously screen for spinal muscular atrophy and severe combined immunodeficiency. Taylor JL, Lee FK, Yazdanpanah GK, Starapoli JF, Liu M, Carulli JP, Sun C, Dobrowolsk SF, Hannon WH, Vogt RF. Clin Chem 2015 Feb; 61 (2): 412-9. Doi: 10.1373/clinchem.2014.231019
Khunti N, Khunti N, Khunti K. 2019. Adherence to type 2 diabetes management. Br J Diabetes. 19:99-104










