Initiating Early Access Programs: 5 Things to Consider
The specific considerations for launching an Early (or Expanded) Access Program in Europe.

Early Access Programs (EAPs) — known as Expanded Access Programs in the US — allow pre- approval access to medicines for certain patients. As well as the patients who benefit from earlier access to potentially life-saving medicines, other stakeholders can also benefit from EAPs.
Physicians are given another option to offer their patients, and gain valuable early experience with a product. Payers have the opportunity to evaluate outcomes in a real-world setting outside of clinical trials, and gain an insight into potential value of the drug. Pharmaceutical companies have good launch preparedness, with the optimal operational processes such as supply chain established, and price guidelines are set for future reference, which may help to facilitate the launch pricing and reimbursement negotiations in the future.
If the EAP is successful, the physicians involved become early adopters of, and believers in, the product, relationships are established, and there is a degree of good will formed with physicians and patient groups alike. All of which can help to optimize market penetration when the product is fully launched, ultimately allowing more patients to benefit. But what are the specific considerations for launching an EAP in a region as diverse as Europe?
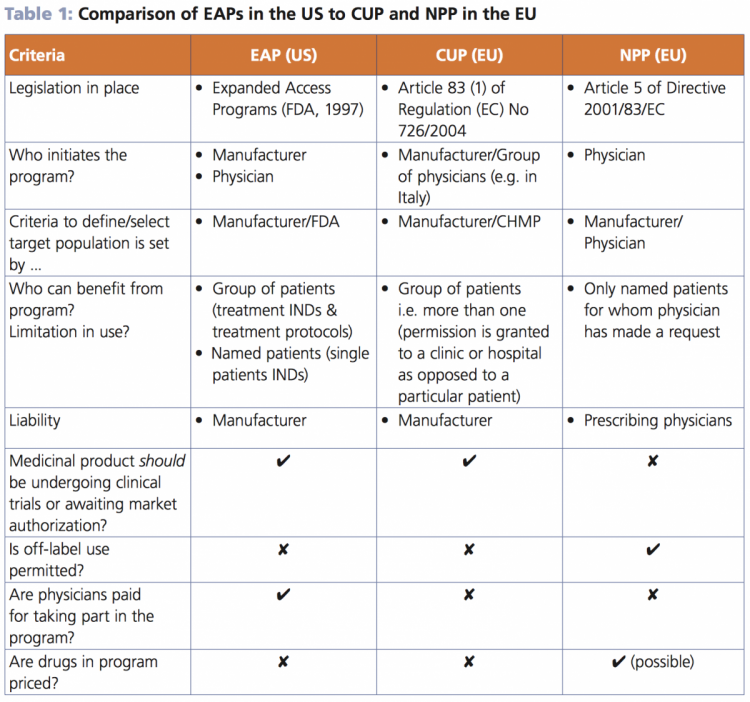
1. EAPs in US vs EU: Differences in regulation and implementation
While the name of EAPs varies by country, there are two main types of early access program in Europe; Compassionate Use Programs (CUPs) and Named-Patient Programs (NPPs). Both of these differ in certain ways to typical Expanded Access Programs in the US:
Compassionate Use Programs (CUPs)
A CUP is the most similar to the typical US Expanded Access Program. A medicinal product is made available for compassionate reasons to a group of patients in a selected clinic or hospital which treats patients with a serious debilitating disease, or where patients whose disease is considered to be life- threatening and who cannot be treated satisfactorily by an authorized medicinal product receive care. In most European countries CUPs are initiated by the pharmaceutical company. Unlike Expanded Access Programs in the US, physicians receive no remuneration for taking part.
Named-Patient Programs (NPPs)
NPPs involve pre-approval access to drugs in response to requests by physicians on behalf of specific, or “named”, patients before those medicines are licensed in the patient’s home country. Whereas CUPs (and Expanded Access Programs in the US) allow physicians to offer the drug to several patients who fulfill the criteria, NPPs are limited to the requested named patient or patients only. Unlike CUPs, NPPs are entirely initiated by physicians, who also bear the liability for it.
Differences in physician remuneration and liability make EAPs in EU more challenging to implement than in the US. On the other hand, the possibility to set a price is an attractive difference in light of future negotiations.
2. EAPs in Europe
There is no ‘one size fits all’ approach in Europe. Individual national state laws, where they exist, may greatly differ from that of European Union and from one country to another, and some countries have more significant barriers to EAPs than others.
Most countries have NPPs in place, but only a few have programs to allow cohorts of patients rather than individuals, to benefit from early access (as in the CUP model).
There are several factors pharmaceutical companies should consider when evaluating which countries within Europe to implement an EAP. These include the degree of administrative effort required to initiate the program (eg, timing required to assess the application and patient volume restrictions), and the feasibility of establishing an initial price for the product through the EAP.
Based on such criteria, the below ‘heat map’ demonstrates the relative degree of difficulty in implementing EAPs (in this case Named Patient Programs) in several European countries - the darker the color, the fewer barriers exist in that country.
France has the fewest barriers – it is the pioneer in Europe for EAPs, and the only country where systematic review and funding of EAPs exists, through the ATU (authorization temporaire d’utilization) system, which is social security-funded and covers both NPPs and CPUs.
Switzerland is also an interesting option, due to the possibility to set a price, the fact that most of the drugs are reimbursed, and that promotion is possible.
Conversely, countries such as Greece and Portugal do not have the infrastructure in place to support EAPs and thus the barriers to implementation are much higher.
Selecting the right countries with highest feasibility for EAP implementation is critical for success and efficient use of resources.
3. EAPs in acute and primary care disorders
There are a number of chronic, debilitating conditions — for example cancer and infectious diseases — which particularly fulfill the criteria for EAPs. But EAPs are not limited to these therapy areas. As an illustration of the therapy areas most commonly associated with EAPs, the graph shows the number of drugs per therapy area which have been granted NPP status in France between 2004 and 2011.
Highlighted in brown are the drugs that received NPP status in the area of cardiology, which include e.g. Argatroban (anti-coagulant), Capoten (hypertension), Simdax (acutely decompensated heart failure) and Syprol (hypertention).
4. Planning timeframes
The timing of EAPs is critical. If the program begins too early, supplies of the drug may not be adequate to meet the needs of both the program and any on-going clinical trials. If the program is started too late, too few patients may be served to justify the effort.
The ideal timing for implementation is usually around 12–18 months prior to launch.
Typically, EAPs involve products that are in Phase III, although planning can begin during Phase II, and should include all relevant stakeholders where possible. The planning element is absolutely essential and should be factored into the timing, allowing for time to prepare documents and contracts, assemble educational materials and establish treatment criteria.
Right timing and advanced planning are important to ensure the EAP runs smoothly and that the patients can benefit as early as possible.
5. Cross-functional teams
Internal buy-in is essential and usually there needs to be cross-functional support for the program to work. This should include involvement from medical affairs, regulatory, marketing and market access (including P&R stakeholders) and supply chain management.
The complexity of conducting EAP feasibility analysis and the implementation itself requires advanced strategic planning to ensure goals are met while avoiding unnecessary hurdles. Strong understanding of the criteria for different types of EAPs, and knowledge of local regulations across different countries, is vital.
If gaps in knowledge exist, for example around some of the specific dynamics in different European countries, some pharmaceutical companies may choose all the implications on their product life-cycle before deciding to start the program. by external expertise to fill knowledge gaps may be the optimal approach. Market Access Roadmap to work with specialized healthcare consultancies who are experts on the design and implementation of such programs in Europe.
Final thoughts
The decision whether to implement an EAP is a strategic one. Pharmaceutical companies should consider all the implications on their product life-cycle before deciding to start the program.
Many in the pharmaceutical industry believe EAPs will be too risky, too complicated or too costly to consider, but if strategically planned and tightly focused, the benefits outweigh the risks.
EAPs have great potential in some European countries to benefit all stakeholders involved, from the patient who receives the medicine early, to the pharmaceutical company who provides it. Although EAPs can represent a significant undertaking, companies who invest in them should see considerable benefit in terms of launch preparedness, relationship building and market penetration.
A proactive strategic plan and a solid understanding of the process and potential barriers involved is essential, as is knowledge around the specific dynamics of each European country. A multi- faceted internal team bolstered by external expertise to fill knowledge gaps may be the optimal approach.










