Thriving in the post-COVID world in 2021 and beyond
How COVID-19 is accelerating existing healthcare trends and the implications for pharma

Introduction
“It's taken this crisis to push us to a new frontier, but there's absolutely no going back.” (by Rick Lee, Executive Chairman, Healthy Platforms and CancerLife)
This quote, assessing the impact of COVID-19 on the cancer care delivery ecosystem1, also nicely encapsulates the impact of the pandemic on healthcare overall. Many of the novel approaches introduced in ultra-rapid timeframes to help under-pressure healthcare systems cope during a state of emergency, are likely here to stay. Many of these approaches are not necessarily new, but rather existing trends that have been adapted and considerably accelerated by the pandemic.
Take the uptake of telehealth / telemedicine for example. It’s an approach that several countries had been tentatively exploring, but during the pandemic, its uptake has skyrocketed. In the US for example, consumer adoption of telehealth has soared, from 11 percent in 2019 to 46 percent today.2
According to one McKinsey article, providers are seeing anywhere between 50 to 175 times the number of patients via telehealth than they did before and approximately $250 billion of US healthcare services could potentially be virtualized.3 It’s just one example. The situation in 2020 has provided the proof of concept for many such novel approaches – now the toothpaste is out of the tube, it will not be going back in.
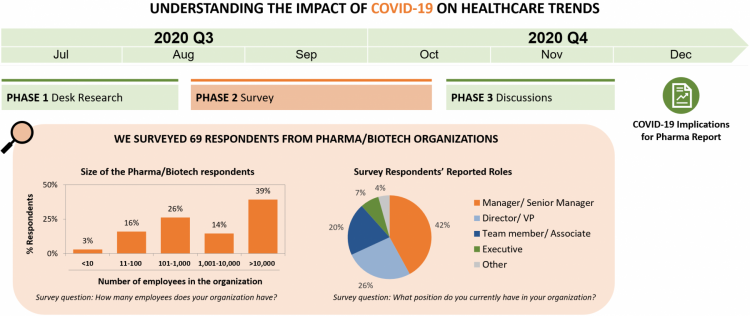
So, what does this mean for the pharmaceutical industry? Which trends do pharma companies believe will accelerate most in the next year? Which ones will impact their companies the most in that time? To answer these questions, we conducted a survey of 69 respondents from pharmaceutical or biotech companies (more information on the survey respondents can be found on page 14).
From initial research, we identified a total of 15 healthcare trends, which may in some way have been accelerated or impacted by COVID-19. The 15 trends can be divided over five key categories: governance, funding, delivery, consumers and operations (the full list of identified trends can be found on page 14).
Survey respondents were asked to rank their top three trends for two key questions – which trends will accelerate the most by 2021, and which will impact pharma the most in 2021.
The results reveal that while it is factors around healthcare delivery and governance that are accelerating most quickly (telehealth being a prime example), the factors that will have the greatest impact for pharma are around healthcare operations and, perhaps unsurprisingly, funding.
In this report we present the findings around these two important questions and explore what impact the leading selected trends are already having and are likely to have in the future – for healthcare systems and pharma companies alike.
Accelerating healthcare trends
Survey participants were asked which 3 of the 15 healthcare trends will accelerate most by 2021. The top 6 responses are in figure 1 below.
Figure 1: Top 6 healthcare trends to accelerate most by 2021

Specialization of care and unlocking clinicians’ capacity
This was the trend that most respondents selected. Certainly, in a time necessitating unprecedented responses from healthcare systems to respond to the crisis, the need to maximize the potential from physician capacity has been paramount.
“Nature Medicine” published a paper4 reviewing some of the “Fast and frugal innovations in response to the COVID-19 pandemic” in 2020, which includes examples of transformation of existing operating infrastructure and repurposing of human resources.
Intensive care units (ICUs) for example, have been reorganized to ensure that more rapid, expanded care is provided to a far larger number of patients. This has required lower-level health workers to handle the preparation of patients in groups, with specialists then intubating one patient after another in rapid succession.
Meanwhile, community health workers can be upskilled to provide household-level advice and support during the epidemic, providing “a distributed public-health response where it is needed most: in households.” The paper cites examples of community health workforces in Brazil, Ethiopia, India and Pakistan that have enabled effective responses during Zika, Ebola and cholera epidemics.
Such examples of “frugal innovation” – that is, doing more, with fewer or existing resources – has been the reality of the experience of many low- and middle-income countries, even before the COVID-19 pandemic. For example, over the last decade, hospitals in India have found ways to provide world-class healthcare at affordable prices through frugal innovation.5 By employing a “hub and spoke” configuration of assets – where spoke facilities channel patients from remote locations to urban hubs for more sophisticated procedures and surgery – patients are managed in larger volumes by more specialized healthcare professionals. Low cost healthcare workers meanwhile have taken over many of the more basic tasks, freeing time for specialists. General physicians are encouraged to become specialists and specialists to become super-specialists.
Such optimization of existing resources is essential in a situation where time and resources are lacking, but is also common sense for longer-term planning for health systems already under huge financial pressure. It is very likely that we will continue to see such attempts to increase operational efficiencies intensify over the year ahead.
Regulation on telemedicine, interoperability and data protection
As stated previously, the boom in telemedicine during the pandemic has been extraordinary. This has been made possible because of unprecedented rapid change to regulations around telemedicine and data protection, the second most often selected trend in our survey.
There is perhaps no better example of this unprecedented change than the US. Before COVID-19, the benefits and available evidence surrounding telehealth “was regarded with skepticism by MedPAC and Congress”.6 There were strict regulatory measures in place, creating a barrier to telehealth and leading to low uptake. COVID-19 changed all of that, almost overnight.
Medicare program requirements were waived in order to accelerate the use of digital medical technologies including telehealth, telemedicine, and remote services for purposes of medical continuity as well as to manage patients who do not require in-patient emergency services. Over 1,000 waivers were put in place to alleviate requirements that would otherwise inhibit payment to providers using telehealth. Although these were temporary, as “The National Law Review” pointed out, “it will be difficult to unwind some of these measures once the COVID pandemic is resolved7,” and there have been moves to make these waivers permanent.
According to a “BMJ” opinion article, the COVID-19 public health emergency has “provided an unprecedented opportunity to evaluate telehealth services and to determine the expanded role they could play in the future of medicine,” and to maximize this opportunity “it will be critical to appropriately modify healthcare policies so that the best aspects of telehealth adoption could be retained.”6
For the US, such an approach was quite a radical about turn, but for other countries – such as Germany – it was a path they were already taking. For several years Germany has been paving the way for digital transformation of its healthcare systems, enabling easier access to healthcare and video consultations for patients, and significantly improved data exchange between healthcare providers. But even here, COVID-19 rapidly transformed the landscape.
Before COVID-19, only a maximum of 1 in 5 patients could be treated by a doctor via video consultation and such consultations were limited to 20% of total services provided. During the pandemic, the National Association of Statutory Health Insurance Physicians and the National Association of Statutory Health Insurance Funds lifted this restriction.8
It is a fascinating area and it will be interesting to see what transpires. Most countries had been tip-toeing forwards very carefully with adoption of telehealth – the sudden sprint in 2020 may need time to settle, and it is vital that measures are in place to evaluate quality and resist abuses of the system. But at this stage, it appears the rapid regulatory changes in this area will allow the full promise of telehealth services to be realized.
Healthcare trends impacting pharma
Survey participants were asked which 3 of the 15 healthcare trends will impact pharma the most in 2021. The top 6 responses are in figure 2 below.
Figure 2: Top 6 healthcare trends to impact pharma most in 2021

Reduced / remote customer engagement
Reduced customer engagement has of course been a direct consequence of COVID-19 for pharma companies, with face to face discussions with healthcare professionals (HCPs) reduced to close to zero in all major countries at the height of the pandemic.9
With in-person visits drastically reduced, companies have needed to find other ways to continue engaging effectively with HCPs. Research published in “Pharmaphorum” revealed that while remote communication with HCPs rose in all countries, a lot of it was via channels such as email and WhatsApp, which while fine for providing quick, basic information, is not conducive to meaningful engagement.
The research found that more meaningful engagement between company reps and HCPs via remote videoconferencing significantly increased in 2020, although not to the levels of pre-pandemic face to face engagement.9
Will use of these remote channels persist? 54% of US physicians and 44% of EU5 physicians agree that remote rep interaction will be sufficient in the future.9 The reality is that some kind of combined approach is likely for many, but the past few months have proven that remote interaction is tenable.
It is not just rep interactions either. All interactions with customers have necessarily shifted to digital channels, and we are likely to see omni-channel strategies embraced more widely in the future.
Take product launches for example. When Novartis launched its new lung cancer medicine Tabrecta in May at the height of the global pandemic, it embraced the need to engage via digital channels “in a much more personalized, coordinated and meaningful manner.”10
It was a completely virtual launch, even internally. To reach healthcare professionals in a meaningful way, the company employed rep videoconferencing but also enhanced websites that were considerably more educational than previous launch websites, providing all the resources that interested HCPs needed at their fingertips. They also launched a Twitter livestream as a novel way to provide peer-to-peer education. “We are trying many different things and tactics, and we are learning a lot from these experiences,” said Ameet Mallik, EVP, Head of US, Novartis Oncology, in a “Pharma Boardroom” article.10 It is an approach that may provide a template of sorts for the future.
Increased cost cutting and resource optimization measures
In terms of cost containment, COVID-19 has hugely increased pressure on already stretched healthcare budgets. Healthcare systems were not designed to deal with such large-scale health challenges that affect whole populations requiring urgent mobilization of resources.
A McKinsey survey revealed that CFOs and finance executives of health systems in the US are expecting a dramatic impact on both operating margin and cash flow.11 Even when accounting for federal funding received, COVID-19 has had a negative financial impact on over 90% of health systems surveyed.11
“The Economist” estimated that – counterintuitively – healthcare spending across the world's 60 biggest economies actually fell by 1.1% in US-dollar terms in 2020. This is because the battle against COVID-19 has led to a sharp drop in spending on other conditions, with non-urgent care cancelled and patients avoiding hospitals and clinics.12 For pharmaceutical companies, this inevitably places more pressure on them around the pricing and reimbursement of their medicines.
A review in “L.E.K. Insights” suggested that the impact on pharmaceutical spending will vary by country in Europe, depending on the COVID-19 burden and the existing budget situation, with the impact likely to be particularly pronounced in Spain, Italy, France and the UK.13
The “L.E.K. Insights” review goes on to state that COVID-19 is likely to affect pharma pricing and market access conditions along three key dimensions: health technology assessments (HTAs), pricing, and utilization and supply. With regards to pricing, various cost containment methods will likely be implemented by payers, although this will likely be accelerated versions of those already in place, such as pay for performance models, indication-based pricing or blunter solutions such as list price reductions or mandatory discounts.
What can pharma companies do to prepare for the new environment? Well, the payer environment will continue to differ at the country level – even more so in the wake of COVID-19 with the differing impact levels, healthcare systems and payer responses in each country – so careful national-level assessments will need to be made. Local affiliates may play a greater role in pricing and reimbursement discussions.
It is also important that companies just continue to monitor local cost containment policies and adapt as required. For example, in the UK GPs are being encouraged to continue to use remote video, online and telephone consultations wherever appropriate. In China, procurement is being centralized and efforts are increasing to cut costs further and be even more productive.
Early dialogue with payers will be even more beneficial to establish evidence requirements in a more financially restrictive environment.
Further reading on the trends
On page 14 you will find the full list of healthcare trends with links to articles, videos or infographics on each of them.
What next? Assess, adjust and innovate
So much has changed so quickly in the healthcare space during the pandemic, that for pharma companies it must feel like trying to build the plane while flying – trying to monitor and react to rapidly-evolving world events while simultaneously trying to prepare and stay one step ahead of them.
A considered response can broadly be divided into three key steps: assess, adjust and innovate. Assessing is about fully understanding the new situation, your place within it, and the potential impact on your business. Adjust is fine-tuning existing plans to ensure that your business performance is not negatively affected by the situation. And innovate is about proactively embracing and preparing for the new reality, adopting a strategy that can allow you to thrive in post COVID-19 world. The survey respondents provided examples of what they are doing at each stage:
Assess:
- > Understand new reality & product resilience
- > Plan for disruption scenarios & mitigation strategies
- > Assess readiness & scale up capabilities
- > Map new stakeholder involvement
Adjust:
- > Review product Value Proposition, including solutions for payers
- > Increase efforts to address coverage & access to care
- > Enhance environmental shaping efforts towards value-based healthcare
- > Re-define collaborations
Innovate:
- > Build-up remote engagement capabilities (also for hybrid reps)
- > Integrate virtual/ digital channels better (omni-channel capabilities)
- > Pilot new digital opportunities for care delivery, including telehealth & AI
- > Develop integrated care/ patient-centric service models
Interestingly, when asked which stage their company is primarily focusing on currently, the largest proportion of respondents (43%) said “Innovate”, although we must acknowledge there may be an element of bias in the evaluation of employees about the progress of their own company.
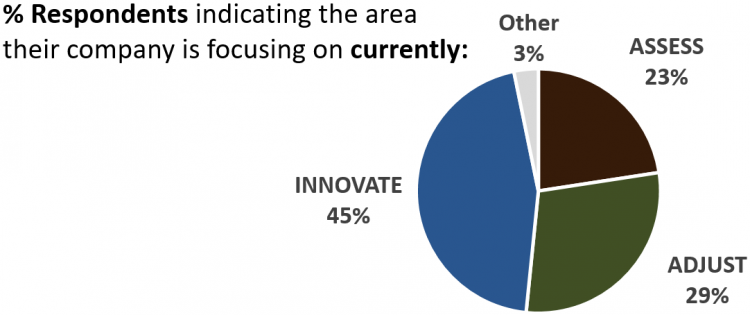
The number for “Innovate” leapt up to 68% when respondents were asked about the main commercial focus of their company in 2021.
Where to play and how to win
But how should companies innovate in a post-COVID-19 world? Executive Insight recommends Pharma companies should redefine their strategies on ‘where to play’ and ‘how to win’ to thrive in this environment. Assessing ‘where to play’ could include redefining your portfolio, asset and service strategy towards more targeted and better serviced patient centric products. It also probably means planning for collaboration with new players who can offer something new. A collaborative approach will ultimately be beneficial to all stakeholders, all of whom have been under immense pressure in 2020.
Assessing ‘how to win’ may include redefining customer and patient engagement models, with new go to market strategies and consideration of new communication channels. It is also important to try to shape environment and policy to support reorganization of care delivery and the patient journey, including new approaches to diagnosis and care.
Want to find out how we could help you to navigate through the new reality? Contact us under info@executiveinsight.ch.
Appendix
The 15 healthcare trends
Based on initial desk-side research, we identified 15 key healthcare trends across 5 core categories, which were used as the basis for the survey. For each trend, we conducted additional research and developed a core communication around it, whether an article, video, infographic or something else. Each of these communications can be accessed by clicking on the relevant trend below (only works in pdf version).

Methodology
This report is the final stage of an ongoing initiative investigating the impact of COVID-19 on healthcare trends.
In Phase 1, we conducted desk-side research to identify the 15 key trends (listed on the previous page). In Phase 2, we conducted a survey of 69 respondents from pharma and biotech organizations, with just over half of those (53%) working in “large” organizations (defined as over 1,000 employees) and the remainder working in “small to medium” organizations (under 1,000 employees). Respondents ranged in seniority, from Manager / Senior Manager (42%), Director / VP (26%), Team Member / Associate (20%), Executive (7%) and Other (4%).
References
1. Impact of COVID-19 on Telehealth. (2020). American health & drug benefits, 13(3), 125–126.
2. McKinsey COVID-19 Consumer Survey, April 27, 2020.
3. Bestsennyy O, Gilbert G, Harris A, and Rost J. Telehealth: A quarter-trillion-dollar post-COVID-19 reality? Available online: https://www.mckinsey.com/industries/healthcare-systems-and-services/our-insights/telehealth-a-quarter-trillion-dollar-post-covid-19-reality
4. Harris, M., Bhatti, Y., Buckley, J. et al. Fast and frugal innovations in response to the COVID-19 pandemic. Nat Med 26, 814–817 (2020). https://doi.org/10.1038/s41591-020-0889-1
5. Govindarajan V, Ramamurti R, Govindarajan V. Delivering World-Class Health Care, Affordably Innovative hospitals in India are pointing the way. Harvard Business Review. November2013. Available online: https://hbr.org/2013/11/delivering-world-class-health-care-affordably
6. Jarrin R, Mori M and Krumholz HM. A regulatory response to covid-19: unleash telehealth. BMJ. June 22, 2020. Available online: https://blogs.bmj.com/bmj/2020/06/22/a-regulatory-response-to-covid-19-unleash-telehealth/
7. Khan E. States Move to Make Permanent COVID-19 Telehealth Waivers. National Law Review. December 14, 2020. Volume X, Number 349
8. Gerke, S., Stern, A.D. & Minssen, T. Germany’s digital health reforms in the COVID-19 era: lessons and opportunities for other countries. npj Digit. Med. 3, 94 (2020). https://doi.org/10.1038/s41746-020-0306-7
9. Rickwood S. Time to change the channel? The future for customer engagement models. Pharmaphorum. October 2020. Available online: https://pharmaphorum.com/sales-marketing/time-to-change-the-channel-the-future-for-customer-engagement-models/
10. Ameet Mallik – EVP, Head of US, Novartis Oncology. Pharma Boardroom. 20 July 2020. Available online: https://pharmaboardroom.com/interviews/ameet-mallik-evp-head-of-u-s-novartis-oncology/
11. Broome B. The financial impact of COVID-19 on health systems and how CFOs are responding. McKinsey. 3 June 2020. Available online: https://www.mckinsey.com/industries/healthcare-systems-and-services/our-insights/the-financial-impact-of-covid-19-on-health-systems-and-how-cfos-are-responding
12. Economist Intelligence Unit. Covid-19: the impact on healthcare expenditure. 27 May 2020. Available online: http://www.eiu.com/industry/article/609621044/covid-19-the-impact-on-healthcare-expenditure/2020-05-27
13. Ahnert V, Seiffert C and Brar S. European Drug Pricing and Market Access in a COVID-19 World. L.E.K. Volume XXII, Issue 80. 14 October 2020. Available online: https://www.lek.com/insights/ei/european-drug-pricing-and-market-access-covid-19-world






