Beyond the Pill – A Global Study
Abstract
An increasingly competitive environment, reduced drug pipelines, multiple patent expiries and payers’ increasing demands for outcomes-based evidence are all factors currently presenting challenges to the pharmaceutical industry. Offering value-added services alongside products, as a means to improve health outcomes and provide a competitive advantage, is a strategy currently being explored by many pharmaceutical companies to confront these challenges. The aim of this study is to analyse to what extent such service strategies are and will be integrated into the pharmaceutical business model. The scope of the study is global and concentrates on pharmaceutical companies manufacturing branded products. In all, 59 experts working for the pharmaceutical industry were asked for their perceptions on current and future service strategies within the industry. Most respondents believed that service strategies are becoming increasingly important. However, there are still multiple challenges to overcome. Legal restrictions, difficulties in measuring return on investments, conservative business philosophies and mistrust towards the pharmaceutical industry are factors slowing down the advance of service strategies.

Introduction
Despite the fact that customer focus has long been a guiding principle within the pharmaceutical industry, it has usually been limited to marketing products with a strong customer orientation. The core corporate strategy within the pharmaceutical industry has essentially remained product-centered.(1) However, the challenges the industry is currently facing, such as an increasingly competitive environment, reduced drug pipelines and multiple patent expiries,(2) and payers’ increasing demands for out- comes-based evidence,3 have raised questions around whether product-centered strategies alone are optimal. Integrating ‘‘beyond the pill’’ value-added services as a means to improve health outcomes and provide a competitive advantage is currently being explored by many within the pharmaceutical industry.(4–6) To date there has been limited research available in this particular field. The aim of this study, therefore, was to assess how ‘‘beyond the pill’ service strategies – defined as a strategy for combining service offerings with a product or portfolio of products targeting stakeholder needs along the patient pathway, supporting better health outcomes while at the same time providing a source of competitive advantage – are perceived by the pharmaceutical industry and how they are currently being integrated. A survey among employees working within the industry was conducted to provide this assessment.
Methodology
The explorative study was conducted with a survey created with SurveyMonkey, an online survey software tool. Data were collected through online question- naires covering 29 questions. The questionnaire consisted of the following nine sections:
- General information
- The current situation within the pharmaceutical industry
- Beyond the pill service strategies
- The development and implementation of service strategies
- The involvement of key customers
- Challenges in service development
- Types of services
- Measurement of return on investment
- Final questions
Different question formats were used depending on the topic, including multiple-choice questions, matrices (using the Likert Scale), one ranking question and open questions. Most multiple-choice questions included the answer possibility of ‘‘other’’ to cover all answer possibilities. One conditional question was included, allowing participants who were not directly involved in ‘‘beyond the pill’’ service strategies to miss out questions related to service strategies within their own companies.
All questions, except open ones, were mandatory. The choice of answering ‘‘I do not know’’ was given in questions requiring a certain level of knowledge (e.g. on how services are implemented in the respondent’s company) but not for questions on personal opinions (e.g. on how respondents think services should be implemented). Thus, participants had to state their opinions or perceptions.
As it could not be taken for granted that all participants understood what was meant by the term ‘‘beyond the pill service strategies’’ and to ensure that all respondents had the same understanding of the term, a definition was given in the survey invitation, the introductory text of the survey and in more detail in the survey itself. The definition within the questionnaire was as follows:
Target group of the survey
The questionnaire was targeted at employees of pharmaceutical companies who, within their role, have some level of involvement with service development. As this is a relatively imprecise target group, who could work across a multitude of functions, it was decided to concentrate on professionals working in marketing, sales, business development, market access, product planning or medical affairs.
Email invitations to the survey were sent out to professionals identified within the online network LinkedIn who met these criteria. Additionally, online invitations were posted on professional platforms (LinkedIn, Xing, Pharma-Mkting Forum) and kept as short as possible to keep the readers interest. The postings therefore were only targeted at professionals ‘‘working in pharma’’ and being ‘‘involved in service offerings or the commercialization of products’’.
The survey roll-out
The survey was open from 05 August to 06 September 2013. Email invitations were sent out to 139 contacts that were known to work for pharmaceutical companies within the fields of marketing, sales, business development, market access, product planning or medical affairs. The emails were sent out through the SurveyMonkey online tool and a reminder email was sent out on 19 August. The weblinks were posted on the first day of the survey opening and reminder posts were published one week before the survey closed.
Analysis
The resulting data were collected in anonymous form. Based on the study’s explorative nature and the limited response rate, only descriptive analyses were conducted. Statistical analyses as part of deductive statistics were not predefined or used. (7) All data were collected automatically within SurveyMonkey and exported to IBM SPSS Statistics 19 for further analysis.
Results
The sample
A total of 59 respondents working for the pharmaceutical industry met the target group criteria and their results were included in the analysis which follows.
The majority of respondents working for pharmaceutical companies were working in the fields of marketing (32%) or sales (24%).
Other fields were business development (10%), market access (9%), medical affairs (5%) and new product planning (8%); 12% stated they worked in other areas, which were: Cross-functional areas of marketing, sales and/or business development, health management and politics, competitive strategy or medical education.
In all, 46% of respondents stated they worked on a national level, 28% on a regional level and 26% on a global level. Those working on a national or a regional level specified they worked in the following regions or countries: Europe or parts of Europe (N1⁄45), Germany (4), the United Kingdom (2), Greece (2) as well as Belgium, Netherlands, Switzerland, Russia, Japan, China, Philippines, North Africa and Australia (each mentioned once). Nineteen respondents did not specify their geographical area of work.
Current perceptions
To assess whether the participants felt there was a need for change within the pharmaceutical industry, they were asked to state their agreement on different statements concerning certain challenges and unmet needs within the industry. These included financial pres- sures, market access barriers and the lack of suitability of ‘‘product-centric’’ approaches to pharmaceutical marketing. The results revealed a high level of agreement with most statements:
- 94% believed that rising price pressures require new business strategies
- 94% agreed that unmet needs leave significant opportunities for improved care
- 92% agreed that drugs alone will no longer be the sole source of differentiation
- 91% agreed that new barriers in market access require higher levels of added value
- 83% believed patients will become increasingly informed about their health and treatment options
- 79% agreed that the sustainability of the traditional ‘‘pill alone’’ business model is uncertain and
- 63% agreed that access to healthcare stakeholders has become increasingly difficult.
Participants were also asked to state their opinion on the current and future importance of ‘‘beyond the pill’’ service strategies. Results show that participants believed that service strategies will be more important in 3 years’ time than they are currently. While 33% believe service strategies are crucial now, 52% believe they will be crucial in 3 years from now.
Design and goals of service strategies
The next part of the survey assessed how service stra- tegies are developed and implemented. Overall, service strategies seem to be well-established amongst the respondents’ companies. Asked if their company offers service strategies, a total of 76% said yes, with 13% of the respondents answering ‘‘yes, for all products’’ and 63% saying ‘‘yes, for selected products’’. A total of 17% agreed that their companies ‘‘have plans to develop service strategies’’ and 6% said that there are none in place.
Participants were then asked for the goals their companies pursue with their service strategies. The most popular response (83%) was to ‘‘increase sales and revenues’’, followed by a ‘‘gain in company’s image’’ (66%); to ‘‘protect market shares’’ (62%); and to ‘‘achieve market access’’ (62%); 21% stated ‘‘achieve target pricing’’ was a goal. Of the 17% (8 respondents in total) who mentioned ‘‘other’’ goals, half of them named patient-related benefits (e.g. to achieve ‘‘better health outcomes’’ or to ‘‘save patients’ lives’’).
When asked about the involvement of external partners in service development, 77% of respondents answered that external partners were involved. ‘‘Technology providers’’, ‘‘creative agencies’’ and ‘‘business consultants’’ were each named by 56% of respondents, while 51% named ‘‘health care providers’’. Other external partners named were Patient associations, universi- ties, hospitals, pharmacies, distributors and health care provider associations.
Every respondent except one believed that the therapeutical area has an influence on the extent of customization of a service strategy. When asked to elaborate their response, many replied that the content, complexity and the need for services depends on the nature of the disease. Some stressed the distinction between primary/speciality care as well as acute/chronic and communicable/non-communicable diseases. Differences in the competitive environments of certain therapeutic areas were also cited.
The respondents were asked whether the therapeutic area would have an influence on the choice of the most important audiences for services; 94% of respondents agreed that it would. Elaborating on their answer, some respondents pointed out that the real gate-holders to medicines and reimbursement decisions changed depending on the therapeutic area. It was further stated that some therapeutic areas have a stronger clinical or payer leadership. National regulations (on reimbursement or patient involvement) are a further factor named that could affect the audience selection.
The respondents said their companies offer several kinds of services. The most common types of services among those surveyed were the provision of eductation and training about the product (stated by 81%); raising awareness around the therapy area (66%); helping improved diagnosis (66%); assisting patient adherence to treatment (62%); patient support with living with the disease (57%), facilitating access (51%); optimizing care processes (51%), as well as monitoring (47%) and supporting the treatment choice (40%).
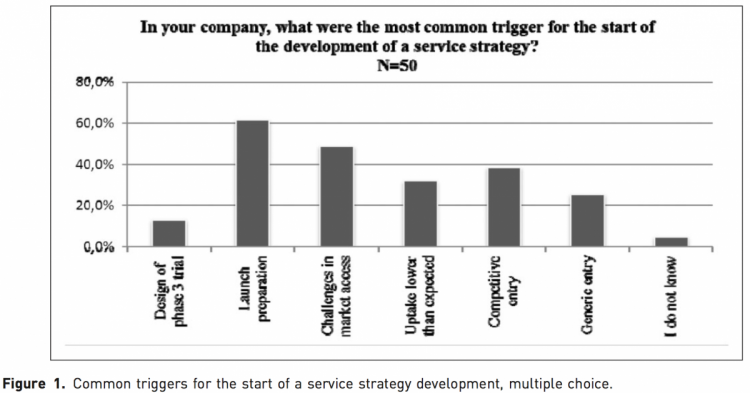
The majority of respondents indicated that the trigger to the development of service strategies happened at some point during the market access process (Figure 1); 62% said that the time of launch preparation was the most common trigger for the start of the development of a service strategy, while 49% said it happened as a reaction to challenges in market access. Service strategies as a reaction to other market challenges (low uptake, competitive and gen- eric entry) were also common responses. Generally it would appear that such strategies are not started at a very early stage in the product lifecycle; the least common starting point out of the options provided was during the design of phase 3 trials.
There was a significant divergence between who the respondents said is currently defining service strategies within their company and who the respondents think should be doing it. A total of 51% stated that service strategies were defined by the marketing department in their company and 32% said by cross-functional team. However, only 22% said the marketing department should do it, while 58% preferred a cross-functional team.
Hurdles in implementation
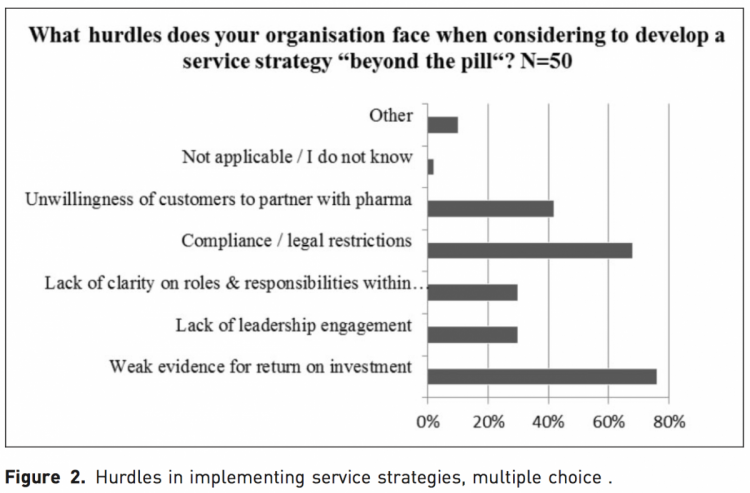
The biggest challenge to service strategy implementation stated by the respondents was weak evidence for return on investment calculations (cited by 76%) as well as legal restrictions (stated by 68%). A total of 42% agreed that the unwillingness of customers to partner with pharmaceutical companies was a barrier, while 30% agreed that a lack of leadership engagement and a lack of clarity on roles and responsibilities within their organization were hurdles. Other hurdles named were that payers or policy makers would not ask for or reward service efforts and that payers’ focus would mainly concentrate on the price of a product. It was also stated that implementing service strategies can be highly complex with a wide time horizon.
Participants were asked to rank the importance of six given metrics to determine the impact of services. According to the survey, the most important metrics for impact measurement were objective outcomes such as increase in diagnostic rates or adherence, followed by an increase in the number of prescriptions. The least important metric was said to be the improvement of company image (Table 1).
Further important metrics for impact measurement mentioned were
- Pharmacoeconomic impact
- Inclusion in guidelines
- Market share gains
- A matrix to reflect the overall treatment paradigm
- rather than just the pill
- Press impact
- Intercompany satisfaction with the project
When asked in an open question what the biggest challenge in determining the impact of a ‘‘beyond the pill’’ service is (Figure 2), many answered the calculation of return on investment (ROI), with the following issues specifically mentioned:
- Difficulties with data access/availability
- Services are not always linked to sales
- Service impact on sales may be difficult to isolate from other effects
- Difficulties in measuring ROI in a specific time period that does not cover all long-term effects
Further specified challenges included the fear of not having positive outcomes and difficulties in the acceptance of key performance indicators by payers and health care providers. One respondent mentioned the scepticism external partners have towards pharmaceutical company involvement, describing it as ‘‘a sliding scale between self-serving and altruistic that skews more towards the former’’.
In a final question, participants were encouraged to share their experiences or comment on ‘‘beyond the pill’’ service strategies.
The importance of fully transparent and trust- worthy collaboration with stakeholders was highlighted:
Similarly, the importance of having clear objectives and ensuring providers understand and share these was also mentioned:
Others criticized the missing focus on patients’ needs:
Discussion and implications
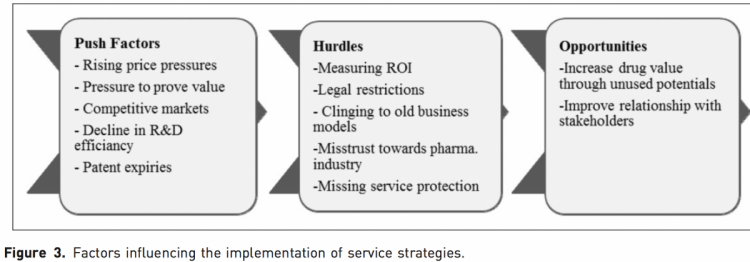
Several trends indicate an increasing importance of new business strategies (Figure 3). Stricter pricing and reimbursement procedures and increasingly com- petitive markets, (1) a decline in drug discovery (8) and patent expiries (4) pose major challenges which the pharmaceutical industry will need to adapt to.
Service strategies have the potential to improve the value of a drug by finding solutions for inefficiencies in the patient pathway and can be included in the value proposition of a product. However, there were also several hurdles identified.
Implications on the implementation of service strategies
Service strategies can be complex in their nature and several different professions within the company will need to be involved. Currently, most services are in the hands of the marketing department, showing its current close link to sales. With a growing sense of importance and acceptance around service strategies, this could change. The service development and implementation could be handled, as preferred by most of the survey respondents, by a dedicated cross-functional team, bringing together different professions like marketing but also medical science liaison or experts in compliance. Important tasks of this cross-functional team would include securing the funding and resources for the project and demonstrating the program benefits for internal decision makers. External partners are already frequently involved in service strategies (as stated by 77% of respondents). They can support service strategies with specialist knowledge and capabilities which may be missing within the company.

Many respondents believe that other stakeholders, particularly patients, should be more involved in the creation of services, but legal compliance is perceived as a major hurdle. This is also reflected in the relatively low numbers who state that patients are actively involved (6%). However, closer collaborations with patients could potentially benefit the service design and acceptance. To ensure such involvement is entirely transparent and compliant, it is vital that compliance professionals are involved in the service design.
Challenges
‘‘Beyond the pill’’ service strategies are still a relatively new concept within the pharmaceutical industry. Currently, internal company structures and pushback within the organizations often due to doubts over ROI, along with a lack of engagement with certain stakeholders, means that the full potential of service strategies are currently not realized, and there are still many hurdles to overcome. The company’s management needs to be assured and convinced of the benefits a service strategy can yield and stakeholders’ trust needs to be gained. One potential solution to provid assurances to pharma management could be the provision of certain incentives in reward for the develop- ment of innovative, high-quality services, whether that could be patent protection for a specified period or an award or some form of recognition. This may help to overcome the ‘‘wait-and-see’’ attitude which many managers currently have, with the concern that the risk of failure might not be worth the possible gains.
However, demonstrating a positive return on investment is the most difficult challenge as indicated by the respondents. Isolating the impact of single services and giving soft outcomes a tangible value were major hurdles named in the survey. Classical return on investment measurement is further complicated by the difficulty of capturing long-term effects, isolating single service effects, accessing sensitive data as well as legal restrictions in profit measurement.
Another significant hurdle named is a lack of trust towards the pharmaceutical industry. Services provided by the industry are suspected to be another hidden form of marketing or to cover hidden price inflations in price negotiations with payers.(9) It might take some time and efforts to achieve stakeholders’ trust, but current trends, such as the industry’s increasing commitment to transparency and the increasing number of private-public-partnerships with the pharmaceutical industry, seem promising.(10)
In addition, services do not need to be exclusively bound to a particular product; they may cross a whole therapeutic area. There are currently several examples of cross-company initiatives offering a joint service base and information.
Generic services could, independent from a product, create new revenue streams for a company and ease the issue of measuring returns on investment. Offering generic services would require new business models, and many companies might not yet be prepared for it. However, other providers may compete on these services, so the window of opportunity is small.
Limitations and future research
The study has been conducted from the point of view of pharmaceutical companies. How services strategies are perceived by stakeholders and providers, to what extent they gain from them and whether the prices for products including service strategies are justified requires separate analysis.
The findings of the survey only represent the opin- ion of a small group within the pharmaceutical industry, and may be biased by selection criteria.
Email contacts were sent out to chosen experts meet- ing the predefined criteria and the online invitations were posted in groups related to service strategies, marketing or sales. The awareness and the perceived importance of service strategies beyond the pill, thus, are likely to be lower in the whole industry compared to the respondents included within the survey.
More extensive and in-depth research is recommended in this field to uncover possible trends both within the industry and within respective stakeholders.
Founding
This research received no specific grant from any funding agency in the public, commercial or not-for-profit sectors.
Conflict of interest
The authors declare that they do not have any conflict of interest.
References
- Baines DA. Problems facing the pharmaceutical industry and approaches to ensure long term viability. MSc Thesis, Pennsylvania: University of Pennsylvania, 2010.
- Mullin R. Beyond the patent cliff. C&EN 2012; 90: 15–20.
- Carone G, Schwierz C and Xavier A. Cost-containment policies in public pharmaceutical spending in the EU. Eur Econ Economic Paper 2012; 1: 1–64.
- Cattell J, Chilukuri S and Knott D. Beyond the pill: creating
- medical value through technology enablement. In: Keeling D and Schrader U (eds) Operations for the executive suite. Opening new horizons for current and future pharma leaders. McKinsey & Company, 2012, pp.85–94.
- Falconi M. At Novartis, the pill is just part of the pitch. The Wall Street J (US edition). 2 January 2013 (accessed 27 March 2014).
- Mattke S, Klautzer L and Mengistu T. Medicines as a service. A new commercial model for big pharma in the postblockbuster world. Santa Monica, CA. RAND Cooperation. OP-381- HLTH. 2012: 1, http://www.rand.org/content/dam/rand/pubs/ occasional_papers/2012/RAND_OP381.pdf (accessed 27 March 2014).
- Fahrmeier L, Künstler R, Pigeot I, et al. Statistik – Der Weg zu Datenanalyse, 7th ed. Heidelberg: Springer, 2010. 8. Scannell JW, Blanckley A, Boldon H, et al. Diagnosing the decline in pharmaceutical R&D efficiency. Nat Rev Drug Discovery 2012; 11: 191–200.
- Ruzicic A and Flostrand S. Valuable collection. What are the prospect for grouping complementary services around a product? Pharmaceutical Market Europe, Jun 2011: pp.32–35.
- Buzzeo B. Achieving Transparency Worldwide: Recent Trends in Global Aggregate Spend and Disclosure Compliance. Cegedim Insights, http://crm.cegedim.com/newsletters/ insights/Documents/AchievingTransparencyWorldwide.html (Oct 2012, accessed 27 March 2014).








